PBRM1 Joins p53 To Impact Kidney Cancer
p53 is a well-known tumor suppressor protein which suppresses cancer by regulating the cell cycle. It is encoded by the gene, TP53. A key component of p53’s suppressor activity is modulated through post-translational modifications, namely acetylation. Acetylation patterns of p53 help determine the protein’s function. These acetylation patterns can be "written," "erased," and "read." "Writing" refers to the addition of acetyl groups by enzymes like histone acetyltransferases, and "Erasing" refers to the removal of acetyl groups by enzymes like histone deacetylases. "Readers" recognize and bind to the acetyl groups on the p53 protein and can inhibit or enhance p53 transcription.
Around half of all cancers have some type of p53 mutation. Clear cell Renal Cell Carcinoma (ccRCC), a common kidney cancer, does not have p53 mutations, leading Cai et al., authors of a Nature Communications paper last year1, to suspect that other mutated genes may be impacting p53 function. Polybromo-1 gene (PBRM1) is mutated in 40% of ccRCC tumors and is required for some p53-mediated processes, so Cai et al. set out to uncover the relationship between these two cancer-associated genes. Could PBRM1 be a reader of p53?
The researchers co-transfected U2OS osteosarcoma cells and human embryonic kidney 293T (HEK293T) cells with both PBRM1 and p53. Next, they induced DNA damage and treated with chemotherapy drugs. Immunoprecipitation showed that PBRM1 binding to p53 was enhanced by DNA damage.
Cai et al. next discovered that PBRM1 binding was diminished when they mutated p53's C terminus domain. Changing acetylation patterns on the C terminus domain using various acetyltransferases enhanced PBRM1 binding. They truncated PBRM1 in series to isolate binding domains and found that, of PBRM's six binding domains, binding domain 4 (BD4) was the only one that bound to p53 by itself. When combinations of binding domains were tested, binding domain 5 enhanced BD4's binding affinity. Subsequent mutation of BD4 abolished the binding affinity.
Now they understood PBRM1 and p53 binding, Cai et al. sought to reveal the impact on function. They knocked PBRM1 out of H1299 cells, human non-small lung carcinoma cells taken from lymph nodes. Twenty-three of p53's downstream targets had diminished expression due to the absence of PBRM1. When tested in kidney cancer cell lines (Remember PBRM1 is mutated in kidney cancer.), PBRM1 knockout followed by DNA damage lead to diminished expression of two very important downstream targets of p53, p21 and MDM2.
What happens to PBRM1 when p53 is knocked out? Again, the expression of downstream targets of p53 are reduced. The binding of p53 and PBRM1 is essential to the tumor suppressing action. When the binding site is mutated, tumor growth increased in cell and mouse models.
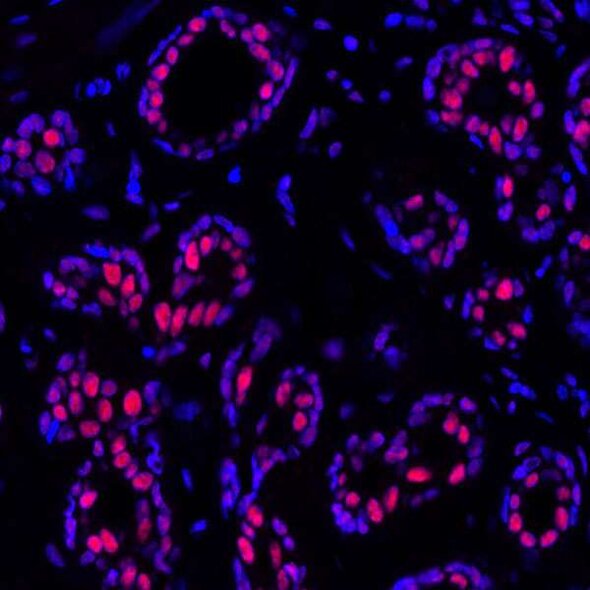
Lastly, the relationship between p53 and PBRM1 was confirmed in human kidney tumors by visualizing PBRM1 and p21 using immunohistochemistry. Staining using antibodies from Bethyl Laboratories against the two proteins revealed a correlation between p21 loss and PBRM1 loss in grade 1 and 2 and stage 1 tumors. Cai et al. hope that these results will contribute to the discovery of effective cancer treatments.
Bethyl offers several PBRM1 antibodies for you research needs.
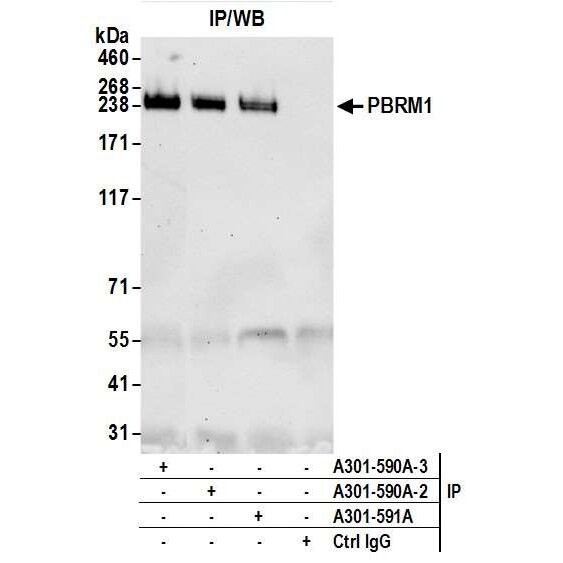
Detection of human PBRM1 by western blot of immunoprecipitates. Samples: Whole cell lysate (1.0 mg per IP reaction; 20% of IP loaded) from HeLa cells prepared using NETN lysis buffer. Antibodies: Affinity purified rabbit anti-PBRM1 antibody A301-590A (lot A301-590A-3) used for IP at 6 µg per reaction. Detection: Chemiluminescence with an exposure time of 30 seconds.
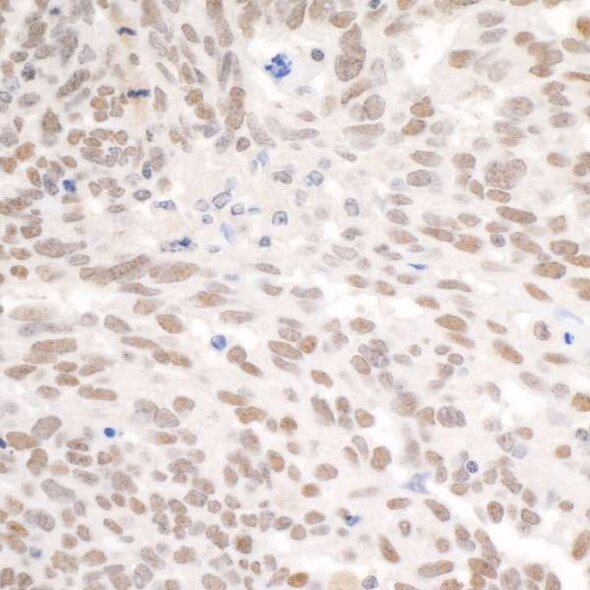
Detection of human PBRM1 by immunohistochemistry. Sample: FFPE section of human ovarian carcinoma. Antibody: Affinity purified rabbit anti-PBRM1 (Cat. No. A301-591A Lot 5) used at a dilution of 1:5,000 (0.2µg/ml). Detection: DAB.
References
- Cai W, Su L, Liao L, Liu ZZ, Langbein L, Dulaimi E, Testa JR, Uzzo RG, Zhong Z, Jiang W, Yan Q, Zhang Q and Yang H. 2019. PBRM1 acts as a p53 lysine-acetylation reader to suppress renal tumor growth. Nat Commun. Dec 20 10(1): 5800.